
U = (–411.3) – (108.7 + 495.Standard enthalpy of formation ( $ \Delta H_f^o $ ) = lattice energy + (2 $ \times $ Electron affinity for chlorine) + Bond Energy of Chlorine gas + first and second ionisation energy of calcium + Sublimation energy of Calcium solid. (hsub for potassium is 89.0 kj/mol, ie1 for potassium is 419 kj/mol, ea1 for chlorine is 349 kj/mol, the bond energy of cl2 is 243 kj/mol, hf for kcl is 436.5 kj/mol. ΔH 4 = Electron affinity of Cl(S)= – 349.0 kJ mol –1 The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Coulomb's law: U k Q1Q2 r0 where U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an ionic solid into the gaseous ions. Use the born-haber cycle to calculate the lattice energy of kcl. Data: Enthalpy of sublimation of Na(s) 107.5 kJ/mol. ΔH 3 = dissociation energy of Cl 2(S) = 244 kJ mol –1 1) Calculate the lattice energy of NaCl(s) using a BornHaber cycle. It has 1:1 stoichiometry ratio of Na:Cl with a molar mass of 58.4 g/mol. 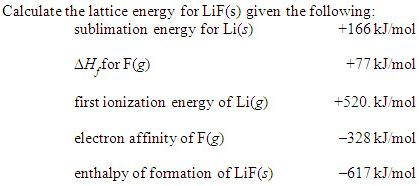
The structure of NaCl is formed by repeating the face centered cubic unit cell. It is extracted from the mineral form halite or evaporation of seawater. ΔH 2 = ionisation energy of Na(S) = 495.0 kJ mol –1 Rock salt ( NaCl NaCl) is an ionic compound that occurs naturally as white crystals. ΔH 1 = heat of sublimation of Na(S) = 108.7 kJ mol –1 ΔH f = heat of formation of sodium chloride = – 411.3 kJ mol –1 Let us calculate the lattice energy of sodium chloride using Born-Haber cycle The sum of the enthalpy changes of these steps is equal to the enthalpy change for the overall reaction from which the lattice enthalpy of NaCl is calculated. Which of the following values most closely approximates the lattice energy of MgO: 256 kJ/mol, 512 kJ/mol, 1023 kJ/mol, 2046 kJ/mol, or 4008 kJ/mol Explain your choice. MgO crystallizes in the same structure as LiF but with a MgO distance of 205 pm. Also, the formation of NaCl can be considered in 5 steps. The lattice energy of LiF is 1023 kJ/mol, and the LiF distance is 201 pm. The bond between ions of opposite charge is strongest when the ions are small. The lattice energy of NaCl, for example, is 787.3 kJ/mol, which is only slightly less than the energy given off when natural gas burns. The lattice energies of ionic compounds are relatively large. Since the reaction is carried out with reactants in elemental forms and products in their standard states, at 1 bar, the overall enthalpy change of the reaction is also the enthalpy of formation for NaCl. Na + ( g) + Cl - ( g) NaCl ( s) Ho -787.3 kJ/mol.  
Let us use the Born - Haber cycle for determining the lattice enthalpy of NaCl as follows: ˆ†H f - enthalpy change for the formation of solid MX directly form elements According to Hess's law of heat summation U - the lattice enthalpy for the formation of solid MX ˆ†H 3- Ionisation energy for M(g) to M +(g) ∆H 4 - electron affinity for the conversion of X(g) to X -(g) ΔH 2- enthalpy change for the dissociation of ½ X 2 (g) to X(g) Here, the energy that must be supplied to 1 mole of sodium chloride to. Born-Haber Cycle is a method used for calculating energy in a chemical reaction.The cycle is primarily concerned with the production of ionic compounds from different elements and involves the formation of ionic compounds from metals (Group I or Group II) when they are reacted with a halogen or a non-metallic element such as oxygen. ˆ†H 1 - enthalpy change for the sublimation M(s) to M(g) calculate the lattice energy as the missing term in the cycle. For example consider the formation of a simple ionic solid such as an alkali metal halide MX, the following steps are considered. Calculate the lattice energy of sodium chloride crystal from the following data: Born exponent, n8, Madelung constant for NaCl1.748, Ionic radius of N a + 0.95 Å, Ionic radius of C l 1.81 Å. The Born–Haber cycle applies Hess's law to calculate the lattice enthalpy. The cycle is concerned with the formation of an ionic compound from the reaction of a metal with a halogen or other non-metallic element such as oxygen.īorn–Haber cycle is primarily used in calculating lattice energy, which cannot be measured directly. It was named after two German scientists Max Born and Fritz Haber who developed this cycle. The Born–Haber cycle is an approach to analyse reaction energies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
